The Significance of Mesenchymal Stem Cells for the Tumor Formation and Progression in Glioblastoma Multiforme
Author
Sloth, Neel
Term
4. term
Publication year
2012
Abstract
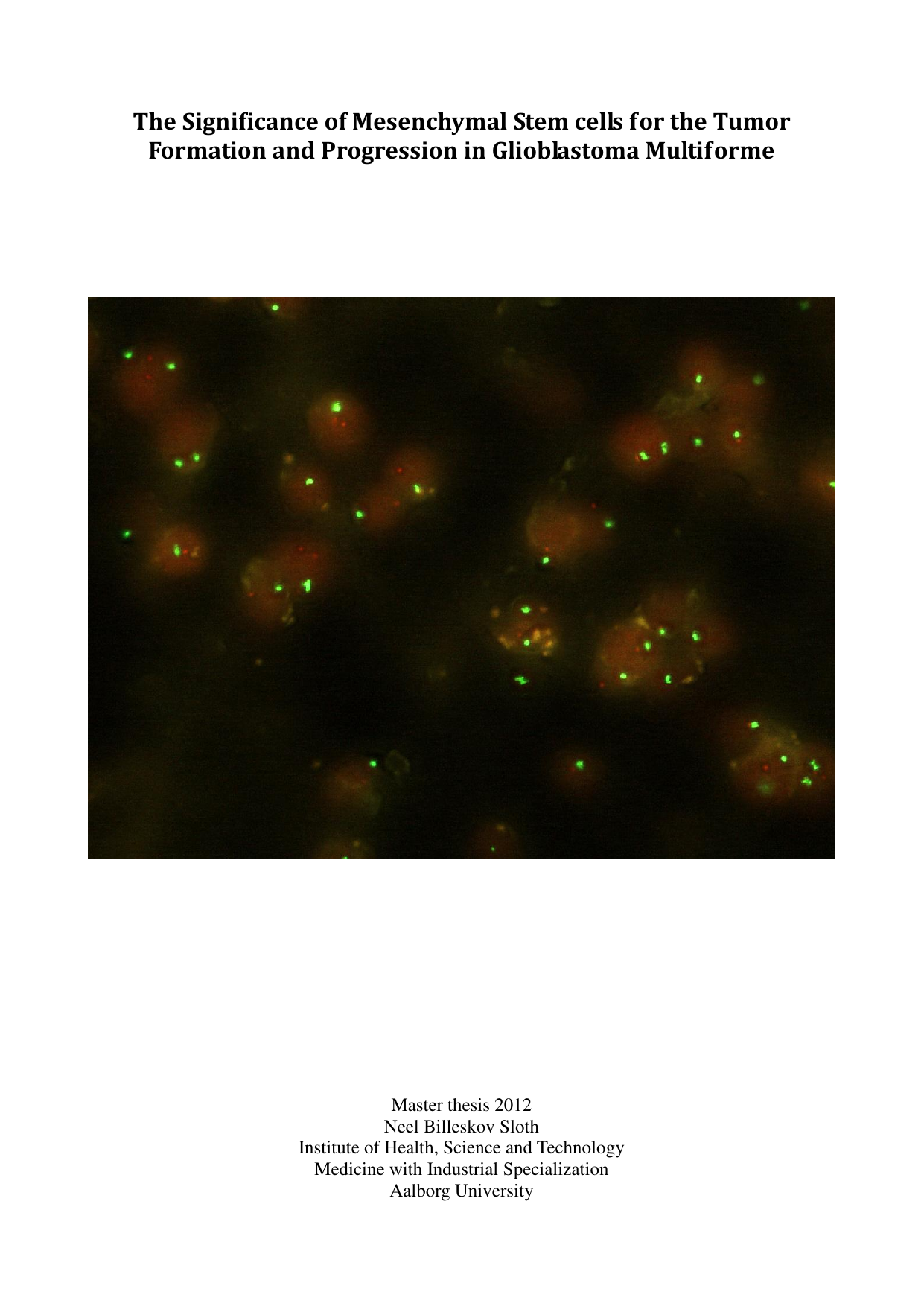
Glioblastoma multiforme is the most aggressive form of brain cancer and carries a very poor prognosis. Mesenchymal stem cells (MSCs) are thought to influence tumor development through mechanisms such as vessel formation, establishment of cancer stem cell niches, promotion of invasiveness, and fusion or transdifferentiation. This thesis combines a literature review of glioblastoma pathology, cancer stem cells, and the potential roles of MSCs with an in vivo experiment in a mouse xenograft model. The aims were to determine the fate of systemically injected adipose-derived stem cells (ADSCs) and to develop an in situ hybridization protocol for tumor sections. In the model, human glioblastoma xenografts were established, ADSCs were delivered systemically, CD105 was selected as an ADSC marker, and tissues were examined by immunohistochemistry and fluorescence in situ hybridization (FISH) with human-specific probes. The results showed successful hybridization of human-specific probes to tumor xenografts, whereas ADSCs could not be detected by immunohistochemistry for CD105 after in vivo injection. It is concluded that pre-labeling cells before injection is more appropriate for in vivo tracking. Overall, the work highlights key technical considerations for studying MSC contributions to glioblastoma progression and can inform future investigations.
Glioblastoma multiforme er den mest aggressive form for hjernetumor og har en meget dårlig prognose. Mesenkymale stamceller (MSC) menes at påvirke tumorudvikling gennem blandt andet kardannelse, etablering af cancerstamcelle-nicher, øget invasivitet samt fusion eller transdifferentiering. Dette speciale kombinerer en teoretisk gennemgang af glioblastoma multiformes patologi, cancerstamceller og MSC’ers mulige roller med et in vivo-eksperiment i en musexenograftmodel. Formålet var at klarlægge skæbnen for systemisk injicerede adipøst-afledte stamceller (ADSC) og at udvikle en protokol for in situ-hybridisering til tumorsektioner. I modellen blev humane glioblastomxenografts etableret, ADSC injiceret systemisk, CD105 valgt som markør for ADSC, og væv analyseret ved immunhistokemi og fluorescens in situ-hybridisering (FISH) med humanspecifikke prober. Resultaterne viste vellykket hybridisering af humanspecifikke prober til tumorxenografts, mens ADSC ikke kunne påvises ved immunhistokemi for CD105 efter in vivo-injektion. Det konkluderes, at forhåndsmærkning af celler før injektion er mere egnet til sporing in vivo. Samlet peger arbejdet på vigtige metodiske hensyn ved undersøgelse af MSC’ers bidrag til glioblastomprogression og kan støtte fremtidige studier på området.
[This apstract has been generated with the help of AI directly from the project full text]
